Neat Info About How To Draw Dot Structures
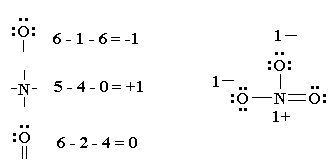
Guidelines for drawing lewis dot structures.
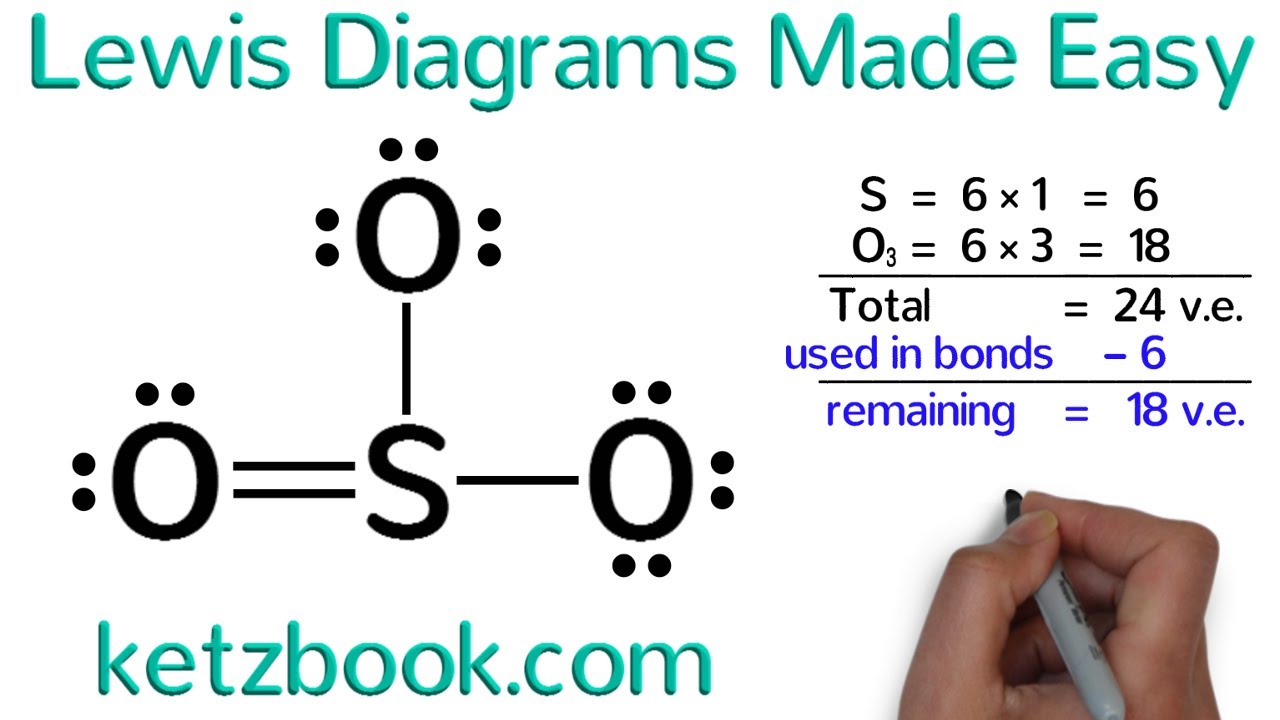
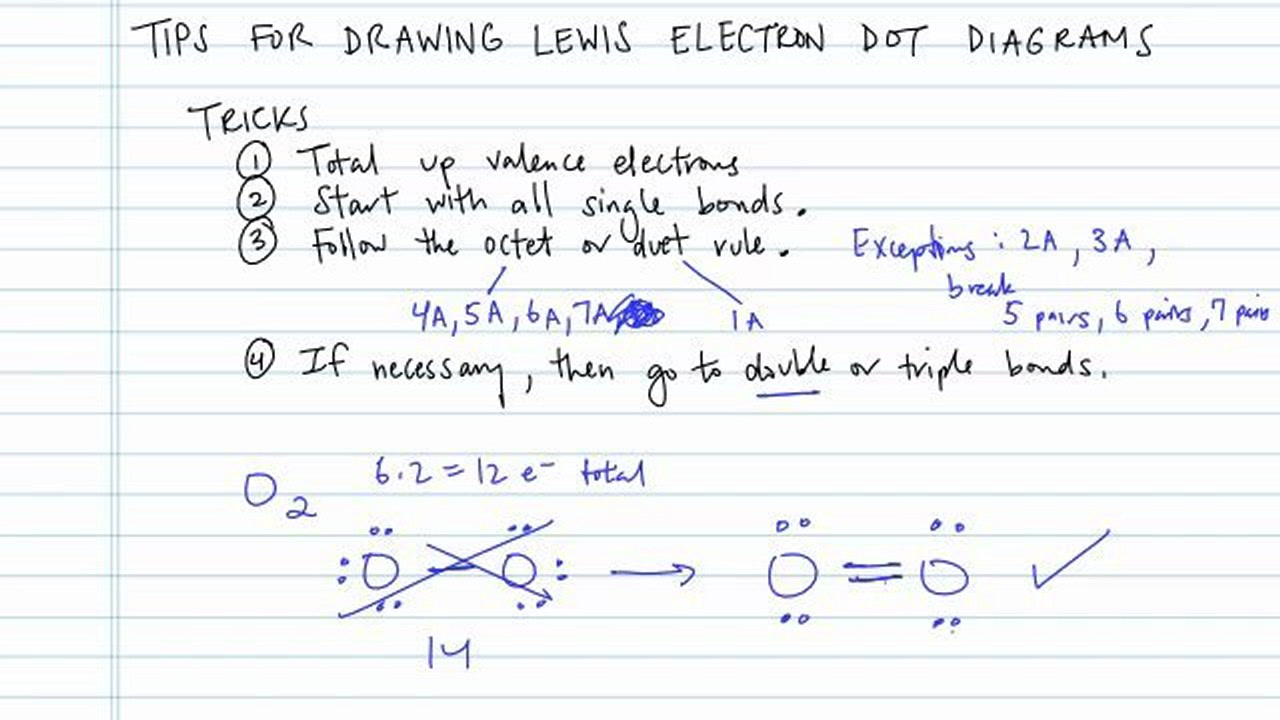
How to draw dot structures. These instructions outline the kelter strategy to draw lewis structures for molecules. Valence electrons are the total number of electrons residing in the. There are several key steps for writing lewis structures:
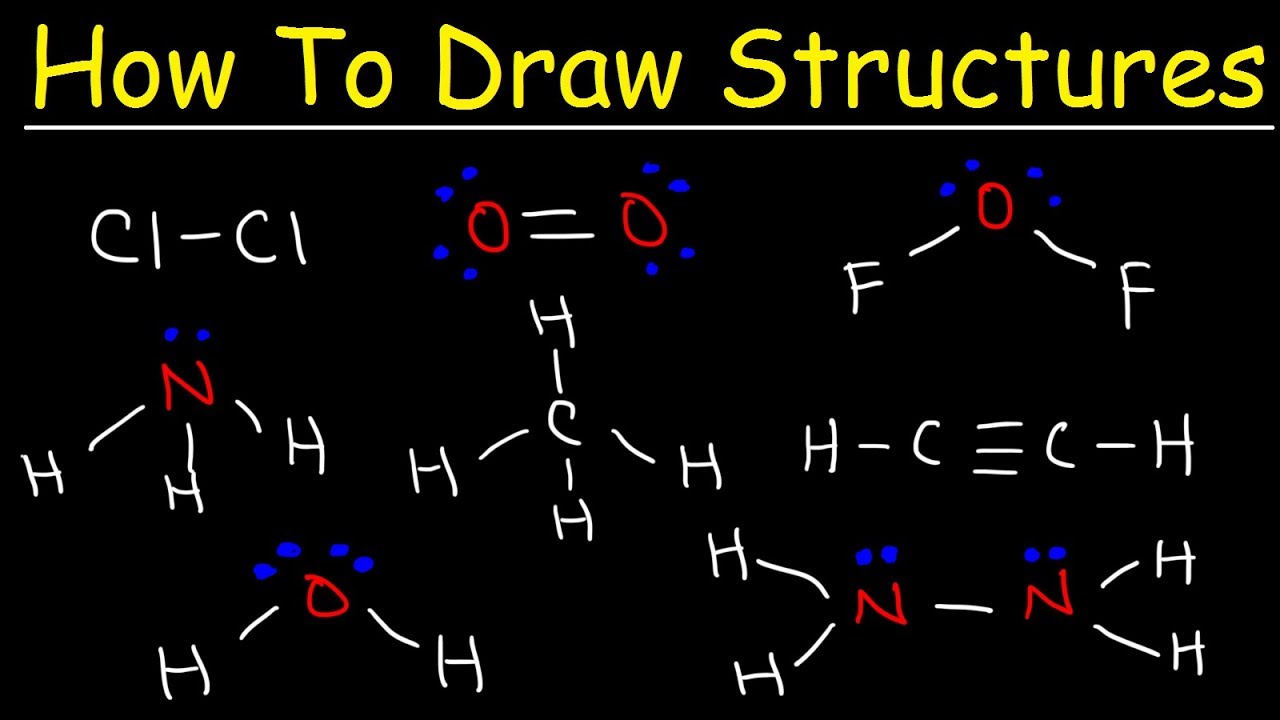
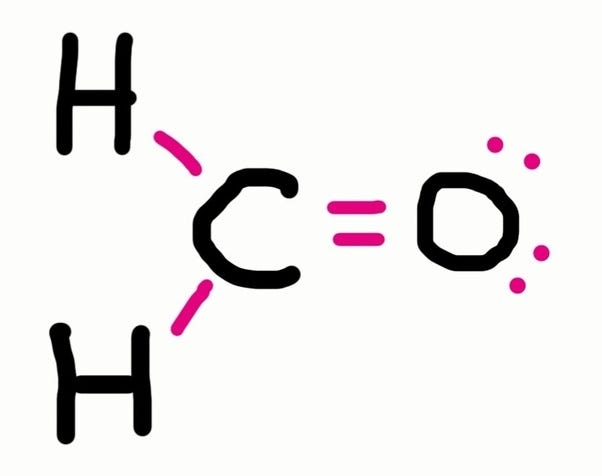
An electron dot structure, also known as a lewis dot formula, is a representation of the electrons found in the valence shell of an atom. A lewis diagram shows how the valence electrons are distributed around the atoms in a molecule. Lewis structure is very important in chemistry, because they are used in.
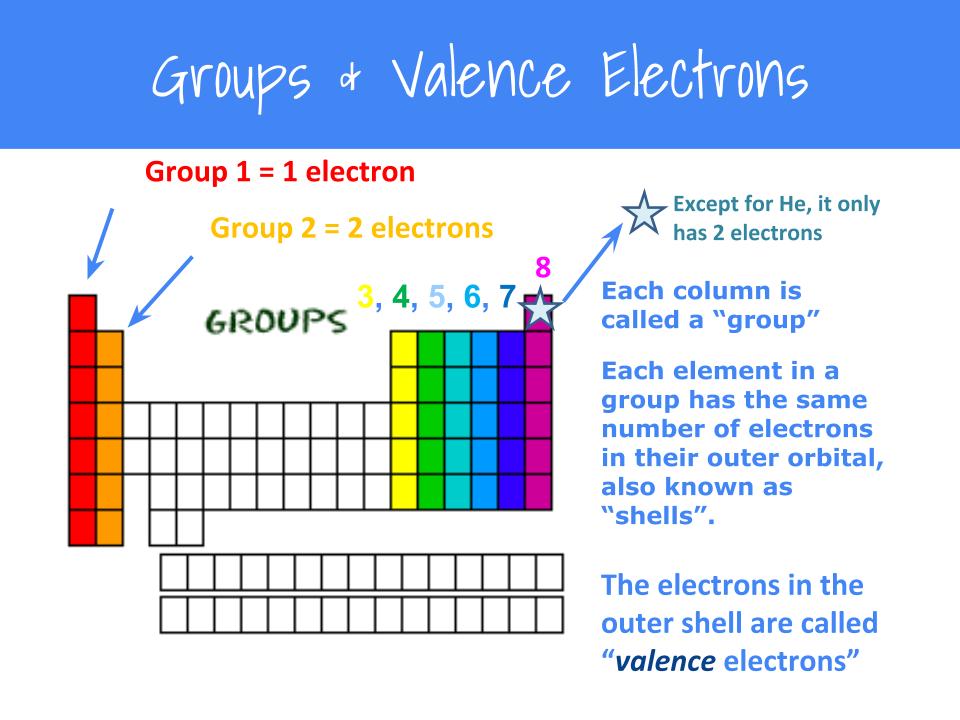
Find the total valence electrons for the molecule. What is an electron dot structure? Electron dot structures or lewis dot formula can be drawn if the molecular formula of the compound is known.
Put the least electronegative atom in the center. Steps to draw a lewis electron dot structure: Lewis dot structure is the classical bonding model in which only valence electrons of the atoms are used.
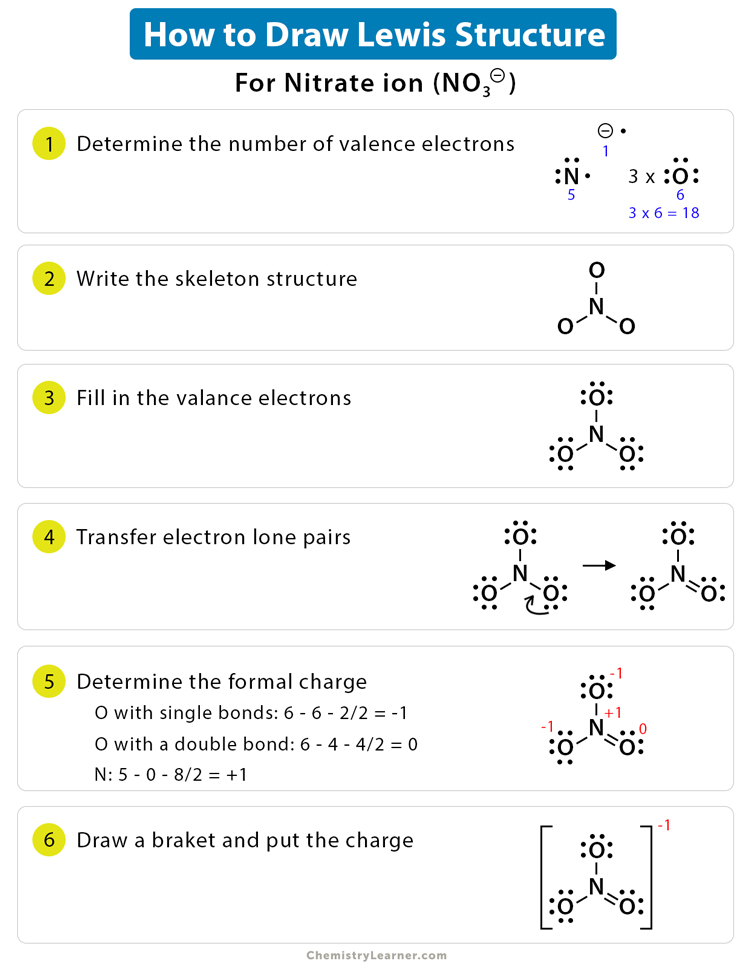
Shared pairs of electrons are drawn as lines between atoms,. Find and count the total valence electrons. To draw the lewis dot structure of positively charged polyatomic ions subtract the positive charge value from the total valence electrons.
Now, we will write down the number of electrons present in each atom of. For example, ammonium ion (nh 4 + ). Hydrogen (h) always goes outside.
Now when you're drawing dot structures, you don't always have to do this step where you're drawing each individual atom and summing all of your valence electrons that way. Find the total valence electrons for the molecule.



/Lewis-dot-structure-58e5390f3df78c5162b4c3db.jpg)